JEE Main | 2014Chemical BondingHard
Question
Among the following oxoacids, the correct decreasing order of acid strength is
Options
A.HOCl > HClO2 > HClO3 > HClO4
B.HClO4 > HOCl > HClO2 > HClO3
C.HClO4 > HClO3 > HClO2 > HOCl
D.HClO2 > HClO4 > HClO3 > HOCl
Solution
HClO4 ⇋ ClO4- + H+
HClO3 ⇋ ClO3- + H+
HClO2 ⇋ ClO2- + H+
HOCl ⇋ ClO- + H+
Resonance produced conjugate base.
(i)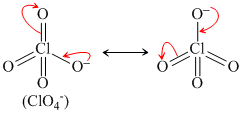
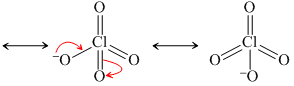
(ii)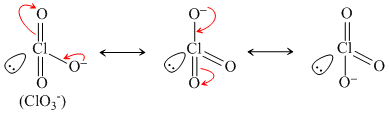
(iii)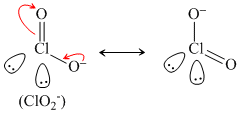
(iv) ClO- is not resonance stabilized.
As per resonance stability order of conjugate base is
ClO4- > ClO3- > ClO2- > ClO-
Hence acidic strength order is
HClO4 > HClO3 > HClO2 > HClO
HClO3 ⇋ ClO3- + H+
HClO2 ⇋ ClO2- + H+
HOCl ⇋ ClO- + H+
Resonance produced conjugate base.
(i)


(ii)

(iii)

(iv) ClO- is not resonance stabilized.
As per resonance stability order of conjugate base is
ClO4- > ClO3- > ClO2- > ClO-
Hence acidic strength order is
HClO4 > HClO3 > HClO2 > HClO
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Among the following moleculesThose having same number of lone pair of Xe are :...The major product in the following reaction is -...Which has Largest value of dipole moment is :-...Given below are two statements:Statement I: The number of species among ${BF}_{4}^{-},{SiF}_{4},{XeF}_{4}$ and ${SF}_{4}...The correct order of increasing X-O-X bond angle is (X = H, F or Cl) :-...