Chemical BondingHard
Question
Which of the following statements is/are wrong ?
The following reactions occur:
i. P4 + 5O2 → P4O10.
ii. P4 + 3O2 → P4O6.
The following reactions occur:
i. P4 + 5O2 → P4O10.
ii. P4 + 3O2 → P4O6.
Options
A.P4 is the limiting quantity.
B.O2 is the limiting quantity.
C.Mass of P4O10 obtained is 2.2 g.
D.Mass of P4O6 obtained in 2.84 g.
Solution
nP4 = 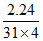 = 0.01 mol, nO2 =
= 0.01 mol, nO2 = 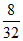 = 0.25 moles
= 0.25 moles
In reaxtion (i), moles of O2 required
= (0.01 mol P4)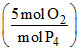
= 0.01 × 5 = 0.05 mol
Since there is more O2 present than required
a. Therefore, P4 is the limiting quanity.
b. Wrong.
c. [Mw P4O10 = 284]
0.01 mol of P4 produces = 0.01 mol of P4O10
= (0.01 mol P4)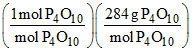
= 0.01 × 284 = 2.84 g
Hence, (c) is wrong.
d. [Mw of P4O6 = 220]
Weight of P4O6 produced
= (0.01 mol P4)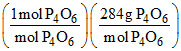
= 0.01 × 220 = 2.2 g
Hence, (d) is wrong
In reaxtion (i), moles of O2 required
= (0.01 mol P4)
= 0.01 × 5 = 0.05 mol
Since there is more O2 present than required
a. Therefore, P4 is the limiting quanity.
b. Wrong.
c. [Mw P4O10 = 284]
0.01 mol of P4 produces = 0.01 mol of P4O10
= (0.01 mol P4)
= 0.01 × 284 = 2.84 g
Hence, (c) is wrong.
d. [Mw of P4O6 = 220]
Weight of P4O6 produced
= (0.01 mol P4)
= 0.01 × 220 = 2.2 g
Hence, (d) is wrong
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
At least one bond pair is situated exactly opposite to another bond pair in -...Correct increasing order for the wavelengths of absorption in the visible region the complexes of Co3+ is :-...The molecule that has linear structure is :...The major product in the following reaction is -...Consider the following sequence of reaction The product B is...