JEE Advanced | 2013Chemical BondingHard
Question
The correct statement(s) about O3 is(are)
Options
A.Thermal decomposition of O3 is endothermic
B.O-O bond lengths are equal
C.O3 is diamagnetic in nature
D.O3 has a bent structure
Solution
(A) 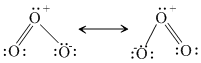
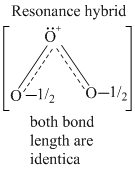
2O3 → 3O2 ; ᐃH = - ve
All the e- are paired in O3 {diamagnetic}
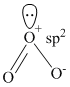
(D) sp2 hybridised ″O″ and bent shape molecule
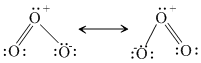

2O3 → 3O2 ; ᐃH = - ve
All the e- are paired in O3 {diamagnetic}
(D) sp2 hybridised ″O″ and bent shape molecule

Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
In HN3 molecule {H - N - N - N} the N - N bond lengths are ............ and ........... from left to right respectively....Which of the following arrangements does not represent the correct order of the property stated against it ?...Give the correct reaction :- (a) NaNO2 NO(g)(b) HNO3 N2O5(c) P4 + NaOH → PH3 + Na3PO4(d) (NH4)2Cr2O7 N2O + Cr2O3 +...Which of the following lanthanoid ions is diamagnetic?(Atoms, Ce = 58, Sm = 62, Yb = 70)...Consider the following structures (A), (B), (C) & (D) Incorrect statement is:-...