Chemical BondingHard
Question
Consider the following structures (A), (B), (C) & (D)
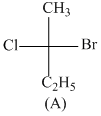
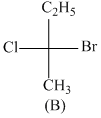
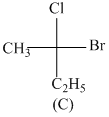
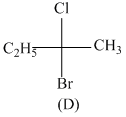
Incorrect statement is:-




Incorrect statement is:-
Options
A.B and C are identical
B.A and B are enantiomers
C.A and C are enantiomers
D.B and D are enantiomers
Solution
B has R configuration and D has R configuration so B and D are identical
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Which of the following has the minimum bound length ?...What is bond enthalpy of XeF bond ?XeF4(g) → Xe+(g) +F-(g) + F2(g) + F(g) ;ᐃrH = 292 KCal/molGiven :I.E. of ...Which of the following structure is similar to graphite?...Which one of the following molecules is expected to exhibit diamagnetic behaviour ?...The spliting of energy level in the presence of a magnetic field is called :...