Chemical BondingHard
Question
Which of the following arrangements does not represent the correct order of the property stated against it ?
Options
A.V2+ < Cr2+ < Mn2 < Fe2+ : paramagnetic behaviour
B.Ni2+ < Co2+ < Fe2+ < Mn2+ : ionic size
C.Co3+ < Fe3+ < Cr3+ < Sc3+ :stability in aqueous solution
D.Sc < Ti < Cr < Mn : number of oxidation states
Solution
V2+ = [Ar] 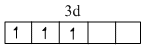
Cr2+ = [Ar]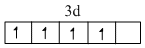
Mn2+ = [Ar]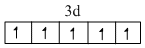
Fe2+ = [Ar]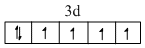
Correct order of Paramagnetic behaviour
Mn2+ > Fe2+ = Cr2+ > V2+

Cr2+ = [Ar]

Mn2+ = [Ar]

Fe2+ = [Ar]

Correct order of Paramagnetic behaviour
Mn2+ > Fe2+ = Cr2+ > V2+
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
In acidic medium, H2O2 - changes Cr2O7-2 to CrO5 which has two (-O-O-) bonds. Oxidation state of Cr in CrO5 is...What is the hybridisation of anionic part of product when XeF2 and PF5 are combined :...The total number of electrons that take part in forming the bond in N2 is :...Dipole moment and ionization constant are maximum in case of :-...In [Ag(CN2)]-, the number of π bonds is :...