Chemical BondingHard
Question
Give the correct reaction :-
(a) NaNO2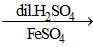 NO(g)
NO(g)
(b) HNO3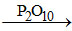 N2O5
N2O5
(c) P4 + NaOH → PH3 + Na3PO4
(d) (NH4)2Cr2O7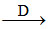 N2O + Cr2O3 + H2O
N2O + Cr2O3 + H2O
Correct option are :-
(a) NaNO2
(b) HNO3
(c) P4 + NaOH → PH3 + Na3PO4
(d) (NH4)2Cr2O7
Correct option are :-
Options
A.a & b
B.a & c
C.b & c
D.b & d
More Chemical Bonding Questions
The geometry of CIO3- according to active to valence shell electron pair repulsion (VSEPR)theory will be...Consider the change in oxidation state of Bromine corresponding to different emf values as shown in the diagram below: B...Which of the following statements is true ?...Which of the following species does not exist under normal condition ?...Which of the following is least likely to behave as Lewis base ?...