JEE Advanced | 2013ThermochemistryHard
Question
The standard enthalpies of formation of CO2(g), H2O(l) and glucose(s) at 250C are -400 kJ/mol, -300 kJ/mol & -1300 kJ/mol respectively. The standard enthalpy of combustion per gram of glucose at 250C is -
Options
A.+ 2900 kJ
B.- 2900 kJ
C.- 16.11 kJ
D.+ 16.11 kJ
Solution
Combustion reaction for glucose is
C6H12O6(s) + 6O2(g)→ 6CO2(g) + 6H2O(l)
áƒHr = 6(-400) + 6(-300) - (-1300)
= -2900 KJ/mol 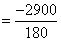 KJ / gm
KJ / gm
= -16.11 KJ/gm
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
Geological conditions are sometimes so extreme that quantities neglected in normal laboratory experiments take on an ove...The Δf Ho for CO2(g), CO(g) and H2O(g) are −393.5, −110.5 and −241.8 kJ mol–1, respectively. The standard enthalpy chang...Calculate the enthalpy of formation (in kcal/mol) of anhydrous Al2Cl6 from the following data.2Al(s) + 6HCl(aq) → Al2Cl6...Combustion of glucose takes place according to the equation :C6H12O6 + 6O2 → 6CO2 + 6H2O, ᐃH = - 72 kcalThe ...Which of the following forms oxide on heating :-...