ThermochemistryHard
Question
Which of the following forms oxide on heating :-
Options
A.LiNO3
B.Li2CO3
C.LiOH
D.All of these
Solution
2LiNO3 → Li2O + 2NO2 + 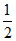 O2
O2
Li2CO3 → Li2O + CO2
2LiOH → Li2O + H2O
Li2CO3 → Li2O + CO2
2LiOH → Li2O + H2O
Create a free account to view solution
View Solution FreeMore Thermochemistry Questions
Reactions involving gold have been of particular interest to a chemist. Consider the following reactions.Au(OH)3 + 4HCl ...The bond energies (in kJ/mol) at 25°C are C–C = 346, C–H = 413, H–H = 437, C = C; 611. From these data, the value of ΔH ...The standard enthalpy of combustion of solid boron is numerically equal to...The enthalpy of combustion at 25°C of H2(g), cyclohexane(l) and cyclohexene(l) are −241, −3920 and −3800 kJ/mol, respect...In an ice calorimeter, a chemical reaction is allowed to occur in thermal contact with an ice water mixture at 0oC. Any ...