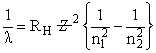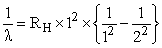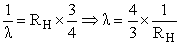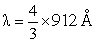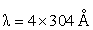Atomic StructureHard
Question
Energy of an electron is given by E = - 2.178 10-18 J 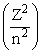 Wavelength of light required to excite an electron in an hydrogen atom from level n = 1 to n = 2 will be :-
Wavelength of light required to excite an electron in an hydrogen atom from level n = 1 to n = 2 will be :-
(h = 6.62 10-34 J-s and c = 3.0 108 m-s-1)
Options
A.
1.214 10-7 m
B.
2.816 10-7 m
C.
6.500 10-7 m
D.
8.500 10-7 m
More Atomic Structure Questions
Which of the following sets of quantum numbers is correct for an electron in 4f orbital?...If there are only two H-atoms, each is in 3rd excited state then :...If the nitrogen atom had electronic configuration 1s7, it would have energy lower than that of the normal ground state c...An α-particle has initial kinetic energy of 25 eV and it is accelerated through the potential difference of 150 vol...Photon having energy equivalent to the binding energy of 4th state of He+ atom is used to eject an electron from the met...