Atomic StructureHard
Question
Which of the following sets of quantum numbers is correct for an electron in 4f orbital?
Options
A.n = 4, I = 3, m = + 4, s = + 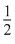
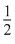
B.n = 3, I = 2, m = - 2, S = + 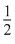
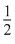
C.n = 4, I = 3, m = + 1, s = + 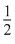
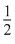
D.n = 4, I = 4, m - 4, s = - 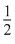
Solution
4f → n = 4
I = 3
m = - l to + l
- 3 to +3
I = 3
m = - l to + l
- 3 to +3
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
It is because of inability of ns2 electrons of the valence shell to participate in bonding that:-...In a certain electronic transition in the hydrogen atoms from an initial state (1) to a final state (2), the difference ...A particle X moving with a certain velocity has a debroglie wave length of 1(Ao), If particle Y has a mass of 25% that o...The total number of orbitals for (n + l) = 4 is...The I.P. of an atom is 16 eV. What is the value of first excitation potential :-...