Gaseous StateHard
Question
For gaseous state, if most probable speed is denoted by C*, average speed by 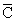 and mean square speed by C, then for a large number of molecules the ratios of these speeds are :
and mean square speed by C, then for a large number of molecules the ratios of these speeds are :
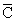 and mean square speed by C, then for a large number of molecules the ratios of these speeds are :
and mean square speed by C, then for a large number of molecules the ratios of these speeds are :Options
A.C* : 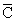 : C = 1.225 : 1.128 : 1
: C = 1.225 : 1.128 : 1
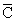 : C = 1.225 : 1.128 : 1
: C = 1.225 : 1.128 : 1B.C* : 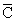 : C = 1.128 : 1.225 : 1
: C = 1.128 : 1.225 : 1
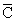 : C = 1.128 : 1.225 : 1
: C = 1.128 : 1.225 : 1C.C* : 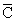 : C = 1 : 1.128 : 1.225
: C = 1 : 1.128 : 1.225
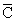 : C = 1 : 1.128 : 1.225
: C = 1 : 1.128 : 1.225D.C* : 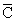 : C = 1 : 1.225 : 1.128
: C = 1 : 1.225 : 1.128
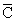 : C = 1 : 1.225 : 1.128
: C = 1 : 1.225 : 1.128Solution
Ump : Uavg : Urms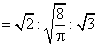
⇒ 1 : 1.128 : 1.224
∴ C* :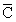 : C = 1 : 1.128 : 1.225
: C = 1 : 1.128 : 1.225
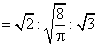
⇒ 1 : 1.128 : 1.224
∴ C* :
 : C = 1 : 1.128 : 1.225
: C = 1 : 1.128 : 1.225Create a free account to view solution
View Solution FreeMore Gaseous State Questions
The compressibility factor of an ideal gas is :...2 litres of moist hydrogen were collected over water at 26oC at a total pressure of one atmosphere. On analysis, it was ...According to Graham′s law, at a given temprerature the ratio of the rates of diffusion of gases A and B is given b...Which of the following is/are correct for an mono atomic ideal gas molecules :...Kinetic theory of gases proves...