Gaseous StateHard
Question
2 litres of moist hydrogen were collected over water at 26oC at a total pressure of one atmosphere. On analysis, it was found that the quantity of H2 collected was 0.0788 mole. What is the mole fraction of H2 in the moist gas
Options
A.0.989
B.0.897
C.0.953
D.0.967
Solution
nTotal = 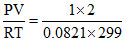 = 0.081 moles
= 0.081 moles
XH2 =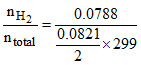 = 0.967
= 0.967
XH2 =
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Calculate the compressibility factor for CO2, if one mole of it occupies 0.4 litre at 300 K and 40 atm. Comment on the r...X mL of H2 gas effuses through a hole in container in 5 sec. The time taken for the effusion of the same volume of the s...The average velocity of an ideal gas molecule at 27oC is 0.3/s. The avarage velocity at 927oC will be :...A real gas obeying Vander Waal′s equation will resemble ideal gas, if the :...Equal masses of methane and oxygen are mixed in an empty container at 25oC. The fraction of the total pressure exerted b...