Gaseous StateHard
Question
The compressibility factor of an ideal gas is :
Options
A.0
B.2
C.1
D.4
Solution
Compressibility factor (Z) is a convenient method of showing deviation of real gasesfrom an ideal gas
Z =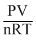
For ideal gas, PV = nRT ; ∴ Z = 1
For ideal gases PV ≠ nRT; ∴ Z ≠ 1
When, Z > 1, refers positive deviation i.e., gas is less compress ible than ideal gas. Z < 1, it refers positive devition i.e., gas is more compressible than ideal gas.
Z =
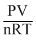
For ideal gas, PV = nRT ; ∴ Z = 1
For ideal gases PV ≠ nRT; ∴ Z ≠ 1
When, Z > 1, refers positive deviation i.e., gas is less compress ible than ideal gas. Z < 1, it refers positive devition i.e., gas is more compressible than ideal gas.
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Which of the following statements are correct ?...A thin balloon filled with air at 47oC has a volume of 3 litre. If on placing it in a cooled room its volume becomes 2.7...Under what conditions will a pure sample of an ideal gas not only exhibit a pressure of 1 atm but also a concentration o...If the pressure of a gas contained in a closed vessel is increased by 0.4 % when heated by 1oC its initial temperature m...For gaseous state at constant temperature which of the following plot is correct ?...