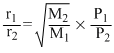Gaseous StateHard
Question
According to Graham′s law, at a given temprerature the ratio of the rates of diffusion of gases A and B is given by:-
(Where P and M are oressures and molecular weights of gases A and B resprctively)
(Where P and M are oressures and molecular weights of gases A and B resprctively)
Options
A.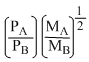
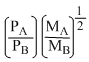
B.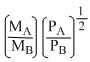
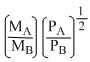
C.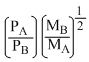
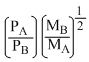
D.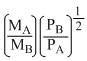
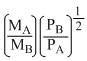
More Gaseous State Questions
The inversion temperature of hydrogen is (given van der Waal′s constants a and b are 0.244 atmL2mol-2 and 1.027 L ...Gas Y + B → milkiness,Gas ′X′ burn with Blue flame then correct choice is :-...1 mol of an ideal gas at 27oC is compressed reverssibly from 20 L to 2L. Find out ᐃS :-...1 mol of a gaseous aliphahatic compound CnH3nOm is completely burnt in an excess of oxygen. The contraction in volume is...If equal weights of oxygen and nitrogen are placed in separate containers of equal volume at the same temperature, which...