Mole ConceptHard
Question
6.02 × 1020 molecules of urea are present in 100 ml of its solution. The concentration of solution is :-
Options
A.0.1 M
B.0.02 M
C.0.01 M
D.0.001M
Solution
0.001M Number of moles 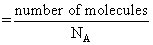
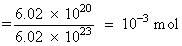
Molar conc.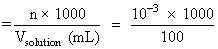
Molar conc. = 0.01 M.
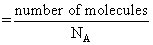
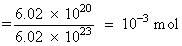
Molar conc.
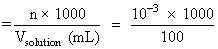
Molar conc. = 0.01 M.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Time required to deposit one milli-mole of aluminium metal by the passage of 9.65 amperes through aqueous solution of al...An element X has three isotopes X20, X21 and X22. The percentage abundance of X20 is 90% and average atomic mass of the ...On repeated sparking 10 mL of a mixture of carbon monoxide and nitrogen required 7 mL of oxygen for combustion. What was...Vapour pressure of chloroform (CHCl3) and dichloromethane (CH2Cl2) at 25oC are 200 mm Hg and 41.5 mm Hg respectively. Va...A metal oxide has the formula M2O3. It can be reduced by hydrogen to give free metal and water. 0.1596 g of the metal ox...