Mole ConceptHard
Question
On repeated sparking 10 mL of a mixture of carbon monoxide and nitrogen required 7 mL of oxygen for combustion. What was the volume of nitrogen ? (All volumes are measured under identical conditions).
Options
A.7/2 mL
B.4 mL
C.7 mL
D.17/2 mL
Solution
CO = x mL, N2 = (10 - x) mL
CO +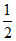 O2 → CO2,
O2 → CO2,
N2 + O2 → 2NO
O2 required =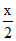 mL, O2 required = (10 - x) mL
mL, O2 required = (10 - x) mL
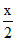 + (10 - x) = 7, x = 6 mL
+ (10 - x) = 7, x = 6 mL
N2 = 10 - 6 = 4 mL
CO +
N2 + O2 → 2NO
O2 required =
N2 = 10 - 6 = 4 mL
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Match the coloum :...In Duma′s method of estimation of nitrogen 0.35 g of an organic compound gave 55 mL of nitrogen collected at 300 K...In the reaction2Al(s) + 6HCl(aq) → 2Al3+(aq) + 6Cl-(aq) + 3H2(g) :-...Change in volume of the system does not alter the number of moles in which of the following equilibria ?...10 g of CaCO3 contains :-...