Mole ConceptHard
Question
The molecular formula of a non stoichiometric oxide of vanadium is VO1.8. The oxide contain both V3+ and V4+ ions. The mole % of vanadium in +3 oxidation state is :-
Options
A.20%
B.60%
C.33.33%
D.40%
Solution
V2O3 + V2O5 = VO1.8
x mol y mol 1 mol
2x + 2y = 1
3x + 5y = 1.8
% of V+3 =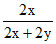 × 100 = 40 %
× 100 = 40 %
x mol y mol 1 mol
2x + 2y = 1
3x + 5y = 1.8
% of V+3 =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
pH of a saturated solution of Ba ( OH )2 is 12. The value of solubility product Ksp of Ba ( OH2 ) is...For the estimation of nitrogen, 1.4 g of an organic compound was digested by Kjeldahl method and the evolved ammonia was...Two samples of HCl of 1 M and 0.25 M are mixed. Find volumes of these samples taken in order to prepare 0.75 M HCl solut...Mole fraction of the Toluene in the vapour phase in equilibrium with a solution of benzene (Po = 120 Torr) and toluene (...The temperature at which molarity of pure water is equal to its molality is :...