Mole ConceptHard
Question
Mole fraction of the Toluene in the vapour phase in equilibrium with a solution of benzene (Po = 120 Torr) and toluene (Po = 80 Torr) having 2.0 moles of each, is -
Options
A.0.50
B.0.25
C.0.60
D.0.40
Solution
PS = PB0XB + PT0XT
= 120 ×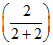 + 80
+ 80 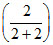
= 100
X′T =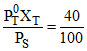 = 0.4
= 0.4
= 120 ×
= 100
X′T =
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
A hydrocarbon C5H8 consumes two moles of hydrogen on catalytic hydrogenation. On ozonolysis, the hydrocarbon produces 2-...Cyclohexanol is dehydrated to cyclohexene on heating with conc. H2SO4. If the yield of this reaction is 75%, then how mu...A gaseous oxide contains 30.4% of nitrogen, one molecule of which contains one nitrogen atom. The density of the oxide r...When hydrocarbons (alkanes, alkenes or alkynes) are burnt completely in excess of oxygen, then...Which of the following statements is/are correct ?20.0 mL of 6.0 M HCl is mixed with 50.0 mL of 2.0 M Ba(OH)2, and 30 mL...