JEE Main | 2014Mole ConceptHard
Question
For the estimation of nitrogen, 1.4 g of an organic compound was digested by Kjeldahl method and the evolved ammonia was absorbed in 60 mL of 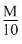 sulphuric acid. The unreacted acid required 20 mL of
sulphuric acid. The unreacted acid required 20 mL of 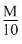 sodium hydroxide for complete neutralization. The percentage of nitrogen in the compound is
sodium hydroxide for complete neutralization. The percentage of nitrogen in the compound is
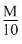 sulphuric acid. The unreacted acid required 20 mL of
sulphuric acid. The unreacted acid required 20 mL of 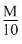 sodium hydroxide for complete neutralization. The percentage of nitrogen in the compound is
sodium hydroxide for complete neutralization. The percentage of nitrogen in the compound isOptions
A.6 %
B.10 %
C.3 %
D.5 %
Solution
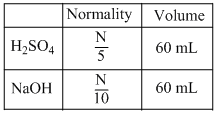
As per question
(ngeq)H2SO4 = (ngeq)NaOH + (ngeq)NH3
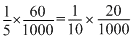 + (ngeq)NH3
+ (ngeq)NH3 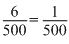 + (ngeq)NH3
+ (ngeq)NH3 (ngeq)N =
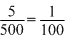
(nmol)N = (nmol)NH3 = (ngeq)NH3 =
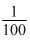
(Mass)N =
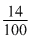 = 0.14 g
= 0.14 gPercentage of ″N″ =
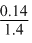 × 100 = 10%
× 100 = 10% Create a free account to view solution
View Solution FreeMore Mole Concept Questions
The pair of species having identical shapes for molecules of both species is...Vapour density of a metal chloride is 66. Its oxide contains 53% metal. The atomic weight of the metal is :-...Which of the following statement(s) is/are correct for water?...Which of the following will have the composition (by mass) as similar as that of acetic acid?...When x g carbon is burnt with y g oxygen in a closed vessel, no residue is left behind. Which of the following statement...