Chemical Kinetics and Nuclear ChemistryHard
Question
A metal crystallizes with a face-centered cubic lattice. The edge of the unit cell is 408 pm. The diameter of the metal atom is
Options
A.288 pm
B.408 pm
C.144 pm
D.204 pm
Solution
For fcc lattice,
4r = √2a
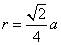
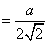
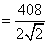
= 144 pm;
diameter d = 2r = 2 × 144 pm
= 288 pm
4r = √2a
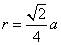
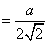
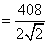
= 144 pm;
diameter d = 2r = 2 × 144 pm
= 288 pm
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The initial rate of hydrolysis of methyl acetate (1M) by a weak acid (HA, 1M) is 1/100th of that of a strong acid (HX, 1...95Am241 and 90Th234 belong respectively to :...Which of the following statements are correct about half-period....For the first order reaction 2N2O5 (g) → 4NO2(g) + O2(g)...The colligative property is not represented by :...