JEE Advanced | 2013Chemical Kinetics and Nuclear ChemistryHard
Question
The initial rate of hydrolysis of methyl acetate (1M) by a weak acid (HA, 1M) is 1/100th of that of a strong acid (HX, 1M), at 25oC. The Ka of HA is
Options
A.
1 10-4
B.
1 10-5
C.
1 10-6
D.
1 10-3
Solution
Rate law for acid catalysed hydrolysis of ester is
rate = k [H+] [ester]
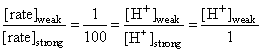
[H+]weak =0.01
HA ⇋ H+ + A-
C-x x x
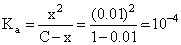
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
For a chemical reaction X → Y, the rate of reaction increases by a factor of 1.837 when the concentration of X is increa...In the acid hydrolysis reaction A + H2O + H+ → Products, where [H+] = 0.1 M and H2O is present in large excess, the appa...The potential energy diagram for a reaction R → P is given below : ᐃH0 of the reaction corresponds to the en...Purple of cassius is colloidal solution of :...For the reversible reaction A$\rightleftharpoons$B, the time at which the rate of change in concentration of ‘A’ is equa...