Chemical Kinetics and Nuclear ChemistryHard
Question
For the first order reaction
2N2O5 (g) → 4NO2(g) + O2(g)
2N2O5 (g) → 4NO2(g) + O2(g)
Options
A.the concentration of the reactant decreases exponentially with time
B.the half-life of the reaction decreases with increasing temperature
C.the half-life of the reaction depends on the initial concentration of the reactant
D.the reaction proceeds to 99.6% completion in eight half-life duration
Solution
For first order reaction
[A] = [A]0e-kt
Hence concentration of [NO0] decreases exponentially.Also, t1/2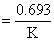 . Which is independent of concentration and t1/2 decreases with the increase of temperature.
. Which is independent of concentration and t1/2 decreases with the increase of temperature.
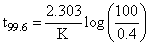
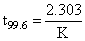 log (2.4) = 8
log (2.4) = 8 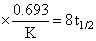
[A] = [A]0e-kt
Hence concentration of [NO0] decreases exponentially.Also, t1/2
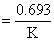 . Which is independent of concentration and t1/2 decreases with the increase of temperature.
. Which is independent of concentration and t1/2 decreases with the increase of temperature.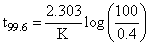
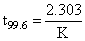 log (2.4) = 8
log (2.4) = 8 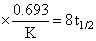
Create a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Milk is colloid in which :...A reaction was found to be second order with respect to the concentration of carbon monoxide. If the concentration of ca...For a chemical reaction X → Y, the rate of reaction increases by a factor of 1.837 when the concentration of X is increa...The correct order of thermal stability of hydroxides is :...Rate law of the reaction A → Product is, rate = k[A]. Graphically it is represented as Hence, rate constant is :...