Gaseous StateHard
Question
Which of the following is not the correct set of pressure and volume at constant temperature and constant moles of gas ?
Options
A.P → 1 atm
V → 200 ml
V → 200 ml
B.P → 760 mm
V → 0.2 L
V → 0.2 L
C.P → 0.5 atm
V → 100 L
V → 100 L
D.P → 2 atm
V → 100 ml
V → 100 ml
Solution
(A) Total moles = 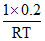
(B) Total moles =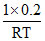
(C) Total moles =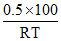
(D) Total moles =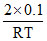
(B) Total moles =
(C) Total moles =
(D) Total moles =
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
A gaseous mixture was prepared by takig equal moles of CO and N2. If the total pressure of the mixture was found 1 atmos...The volume of CO2 produced by the combustion of 40 ml of gaseous acetone in excess of oxygen is...For gaseous state, if most probable speed is denoted by C*, average speed by and mean square speed by C, then for a larg...A gas occupies a volume of 300 cc at 27o Cand 620 mm pressure. The volume of gas at 47o C and 640 mm pressure is :...For the non-zero values of force of attraction between gas molecules, gas equation will be :...