Gaseous StateHard
Question
In the figure-2 the pressure of the confined gas will be
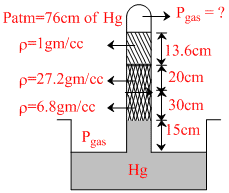

Options
A.30 cm of Hg
B.40 cm of Hg
C.36 cm of Hg
D.46 cm of Hg
Solution
76 × 13.6 = x × 13.6 + 1 × 13.6 + 3.4 × 20 + 6.8 × 30 + 13.6 × 15
76 × 13.6 = x × 13.6 + 13.6 + 13.6 × 5 + 13.6 × 15 + 13.6 × 15
76 = x + 1 + 5 + 30
x = 40 cm
76 × 13.6 = x × 13.6 + 13.6 + 13.6 × 5 + 13.6 × 15 + 13.6 × 15
76 = x + 1 + 5 + 30
x = 40 cm
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
At low pressures (For 1 mole), the Vander Waal′s equation is written as V = RTThe compressibility factor is then e...NH3 gas is liquefied more easily than N2. Hence:...One mole of a non-ideal gas undergoes a change of state (2.0 atm, 3.0L, 95K) → (4.0 atm, 5.0L, 245K) with a change...The order of ionisation potential between He+ ion and H-atom (both species are in gaseous state) is :-...A real gas most closely approaches the behaviour of an ideal gas at -...