SolutionHard
Question
100 ml aq. solution of glucose with osmotic pressure 1.2 atm at 25oC is mixed with 300 ml aq. solution of urea of 2.4 atm at 25oC. The osmotic pressure of mixture is :-
Options
A.3.6 atm
B.1.8 atm
C.1.68 atm
D.2.1 atm
Solution
π1v1 + π2v2 = πR(v1 + v2)
(Resultant O.P.) πR =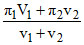
(Resultant O.P.) πR =
Create a free account to view solution
View Solution FreeMore Solution Questions
Name the gas that can readily decolourise acidified KMnO4 solution :...Aqueous solution of barium phosphate which is 100% ionised has ᐃTf / Kf as 0.05. Hence, given solution is...Assuming the formation of an ideal solution, determine the boiling point of a mixture containing 1560 g benzene (molar m...After removing the hard shell of an egg by dissolving in dilute HCl, a semipermeable membrane can be visible. If such an...Which represents correct difference ?I. II. III....