SolutionHard
Question
Which represents correct difference ?
I.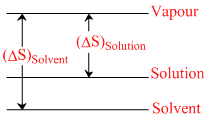 II.
II. 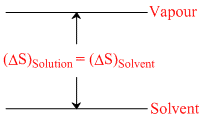 III.
III.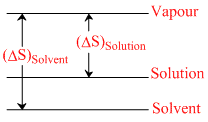
I.
 II.
II.  III.
III.
Options
A.I, II, III
B.I, III
C.II, III
D.I, II
Solution
I : ᐃSsolvent = Svapour - Ssolvent
ᐃSsolution = Svapour - Ssolution
II : ᐃHsolution = ᐃHsolvent = Hvapour - Hsolvent
III : ᐃTb = (boiling point)solution - (boiling point)solvent
ᐃSsolution = Svapour - Ssolution
II : ᐃHsolution = ᐃHsolvent = Hvapour - Hsolvent
III : ᐃTb = (boiling point)solution - (boiling point)solvent
Create a free account to view solution
View Solution FreeMore Solution Questions
1.0 molal aqueous solution of an electrolyte X3Y2 is 25% ionized. The boiling point of the solution is ( Kb for H2O = 0....Which of the following statement(s) is/are true for a colloidal solution of gold in water (Gold sol.) :...A complex is represented as CoCl3.xNH3. It′s 0.1 molal solution in water shows depression in freezing point 0.558 ...The resistance of a solution ′A′ is 40 ohm and that of solution ′B′ is 70 ohm, both solutions be...A teacher one day pointed out to his students the preculia fact that water is unique liquid which freezes exactly at 0o ...