SolutionHard
Question
20 g of a binary electrolyte (mol. mass = 100) are dissolved in 500 g of water. The freezing point of the solution is -0.744oC, Kf = 1.86 K molality-1. The degree of ionisation of the electrolyte is :-
Options
A.50%
B.75%
C.100%
D.Zero
Solution
ᐃTf = i(Kf × molality)
0.744 = i(1.86 ×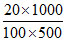 )
)
i = 1
0.744 = i(1.86 ×
i = 1
Create a free account to view solution
View Solution FreeMore Solution Questions
Which of the following liquid pairs shows a positive deviation from Raoult′s law ?...Select correct statement(s) :...When during electrolysis of a solution of AgNO3 9650 coulombs of charge pass through the electroplating bath, the mass o...A mixture of ethyl alcohol and propyl alcohol has a vapour pressure of 290 mm at 300 K. The vapour pressure of propyl al...To 10 mL of 1 M BaCl2 solution 5 mL of 0.5 M K2SO4 is added. BaSO4 is precipitated out. What will happen ?...