SolutionHardBloom L3
Question
A mixture of ethyl alcohol and propyl alcohol has a vapour pressure of 290 mm at 300 K. The vapour pressure of propyl alcohol is 200 mm. If the mole fraction of ethyl alcohol is 0.6, its vapour pressure (in mm) at the same temperature will be
Options
A.350
B.300
C.700
D.360
Solution
Let the vapour pressure of pure ethyl alcohol be P,
According to Raoult′s law
290 = 200 × 0.4 + P × 0.6
P =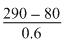 = 350 mm Hg
= 350 mm Hg
Hence, (1) is correct.
According to Raoult′s law
290 = 200 × 0.4 + P × 0.6
P =
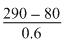 = 350 mm Hg
= 350 mm Hg Hence, (1) is correct.
Create a free account to view solution
View Solution FreeMore Solution Questions
Water should boil at 90°C at pressure...Pure water boils at 373 K and nitric acid at 359 K. The azeotropic mixture of water and nitric acid boils at 393.5 K. On...Which aqueous solution can not be stirred with silver spoon:-...A solution of a non-volatile solute in water freezes at −1.0°C. The vapour pressure of pure water at 298 K is 24.24 mm H...The solubility of Al(OH)3 and Zn(OH)2 are and 1.8 × 10-14, respectively. If NH4OH is added to a solution containing...