Atomic StructureHard
Question
An alphanucleus of energy 1/2 mv2 bombards a heavy nuclear target of charge Ze. Then the distance of closest approach for the alpha nucleus will be proportional to
Options
A.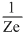
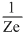
B.v2
C.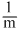
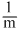
D.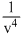
Solution
At the distance of closest approach d,
Kinetic energy = Potential energy
 mv2 =
mv2 = 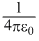
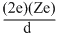
where,
Ze = charge of target nucleus
2e = charge of alpha nucleus
 mv2 = kinetic energy of alpha nucleus of mass m moving with velocity v.
mv2 = kinetic energy of alpha nucleus of mass m moving with velocity v.
or,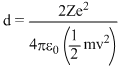 ∴ d ∝
∴ d ∝ 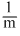 .
.
Kinetic energy = Potential energy
 mv2 =
mv2 = 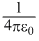
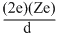
where,
Ze = charge of target nucleus
2e = charge of alpha nucleus
 mv2 = kinetic energy of alpha nucleus of mass m moving with velocity v.
mv2 = kinetic energy of alpha nucleus of mass m moving with velocity v. or,
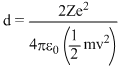 ∴ d ∝
∴ d ∝ 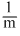 .
.Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
When radioactive substance emits an α - particle, then its position in the periodic in the periodic table is lowere...Starting with a simple of pure 66Cu, of it decays in to Zn in 15 minutes. The corresponding half life is...Which of the following statements is correct?...Number of photons of equal energy emitted per second by a 6 mW laser source operating at 663 nm is $\_\_\_\_$ .(Given : ...A radiation of energy E falls normally on a perfectly reflecting surface. The momentum transferred to the surface is...