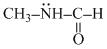Ionic EquilibriumHard
Question
Which of the following the weakest base is :
Options
A.C6H5CH2NH2
B.C6H5CH2NHCH3
C.NO2CH2NH2
D.CH3NHCHO
More Ionic Equilibrium Questions
The solubility product of CaF2 is 1.08 × 10−10. What mass of CaF2 will dissolve in 500 ml water in order to make a satur...Aniline behaves as a weak base. When 0.1 M, 50 ml solution of aniline was mixed with 0.1 M, 25 ml solution of HCl the pH...pOH of H2O is 7.0 at 298 K. If water is heated at 350 K, which of the following statement should be true?...Which of the following buffers have a pH greater than 7 ?(a) AgOH + AgBr(b) CH3COOH + CH3COONa(c) Mg(OH)2 + Mg(OH)Cl(d) ...The acid ionization constant of Zn2+ is 2.0 × 10−10. What is the basic dissociation constant of Zn(OH)+?...