Ionic EquilibriumHard
Question
Aniline behaves as a weak base. When 0.1 M, 50 ml solution of aniline was mixed with 0.1 M, 25 ml solution of HCl the pH of resulting solution was 8. Then the pH of 0.01 M solution of aniliniumchloride will be (Kw = 10-14)
Options
A.6
B.6.5
C.5
D.5.5
Solution
C6H5NH2 + H+ C6H5NH3+
t = 0 5 2.5
teq 2.5 - 2.5
pOH = pKa = 14 - 8 = 6
Now for the solution of [C6H5NH3+] = 0.01 M
pH = 7 -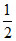 pKa -
pKa - 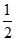 log C = 7 -
log C = 7 - 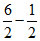 log (0.01) = 5
log (0.01) = 5
t = 0 5 2.5
teq 2.5 - 2.5
pOH = pKa = 14 - 8 = 6
Now for the solution of [C6H5NH3+] = 0.01 M
pH = 7 -
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Calcium lactate is a salt of weak acid and represented as Ca(Lac)2. A saturated solution of Ca(Lac)2 contains 0.125 mole...An amount of 0.10 moles of AgCl(s) is added to one litre of water. Next, the crystals of NaBr are added until 75% of the...Biodegradable polymer which can be produced fromglycine and aminocaproic acid is :-...A salt of strong acid and a weak base is dissolved in water. Its hydrolysis in solution is...Concentrated sulphuric acid is added followed by heating of each of the following test tubes labbelled (I) to (IV) Which...