ElectrochemistryHard
Question
Which statement is correct.
Options
A.A solution of copper (II) sulphate can be stored in iron vessel.
B.An oxide layer on zinc vessel can be easily removed by washing with dilute HCl.
C.Molten PbBr2 is good conductor of electricity because it contains free ions.
D.In the reaction, Li + 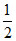 H2 → LiH, hydrogen is a reducing agent.
H2 → LiH, hydrogen is a reducing agent.
Solution
Ionic compounds in molten state are conductor of electricity because of free ions.
Create a free account to view solution
View Solution FreeMore Electrochemistry Questions
Copper can be deposited from acidified copper sulphate and alkaline cuprous cyanide. If the same current is passed for a...Which of the following steyements is true for the electrochemical Daniel cell?...When a lead storage battery is discharged...Consider the following statements:(I) is more stable than (II) is more stable than (III) is more stable than (IV) is mor...Which of the following statements is true for the electrochemical, Daniel cell ?...