Ionic EquilibriumHard
Question
When 20 gm of naphthoic acid (C11H8O2) is dissolved in 50 gm of benzene (Kf = 1.72 Kg/m) a freezing point depression of 2K is observed. The van′t hoff factor is
Options
A.0.5
B.1
C.0.2
D.0.4
Solution
ᐃTf = i × KF × m
2 = 1.72 ×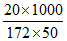 × i
× i
i =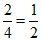 = 0.5
= 0.5
2 = 1.72 ×
i =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Species acting as both bronsted acid as well as base :-...H2CO3 ionizes as follows.H2CO3 $\rightleftharpoons$ H+ + HCO3−; K1 = 4.0 × 10−6HCO3− $\rightleftharpoons$H+ + CO32−; K...Consider an aqueous solution, 0.1 M each in HOCN, HCOOH, (COOH)2 and H3PO4, for HOCN, we can write Ka(HOCN) = . [H+] in ...The molar solubility in terms of solubility product Ksp is given by the relation...Concentrated sulphuric acid is added followed by heating of each of the following test tubes labbelled (I) to (IV) Which...