Ionic EquilibriumHard
Question
Concentrated sulphuric acid is added followed by heating of each of the following test tubes labbelled (I) to (IV)
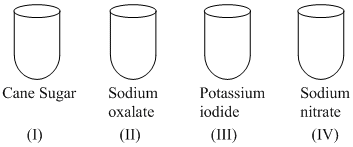
Which of the following statement is incorrect about these observation

Which of the following statement is incorrect about these observation
Options
A.Evolution of violet gas take place in (III) tube and black substance in (I) tube
B.Evolution of neutral and colourless gas take place in (II) tube and neutral gas burn with blue flame
C.Evolution of brown fumes take place in (IV) tube
D.Evolution of violet gas take place in (III) tube and violet gas gives green colour with starch paper
More Ionic Equilibrium Questions
A black sulphide is formed by the action of H2S on:-...Which of the following solutions will have pH close to 1.0?...A buffer solution is prepared in which the concentration of NH3 is 0.30 M and the concentration of NH4+ is 0.20 M. If th...As acidic buffer solution can be prepared by mixing the solutions of :...A cyclic process ABCD is shown in PV diagram for an ideal gas. Which of the following diagram represents the same proces...