Ionic EquilibriumHard
Question
Consider an aqueous solution, 0.1 M each in HOCN, HCOOH, (COOH)2 and H3PO4, for HOCN, we can write Ka(HOCN) = 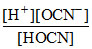 . [H+] in this expression refers to
. [H+] in this expression refers to
Options
A.H+ ions released by HOCN
B.Sum of H+ ions released by all monoprotic acids
C.Sum of H+ ions released only the first dissociation of all the acids.
D.Overall H+ ion concentration in the solution.
Solution
For HOCN- , Ka = 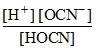 ; Here [H+] = total H+ concentration of solution.
; Here [H+] = total H+ concentration of solution.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
For pure water...In the following reaction HC2O4- (aq) + PO43- (aq) HPO42-(aq) + C2O42-(aq), which are the two Bronsted bases?...To 0.35 L of 0.1 M – NH3 0.15 L of 0.1 M-MgCl2 is added. What minimum mass of (NH4)2SO4 should be added to cause the Mg(...The solubility of PbCI2 in water is 0.01 M 25oC. Its maximum concentration in 0.1 M NaCI will be:...For weak electrolyte, AB, the degree of ionization would be (V = volume of solution having 1 mole of electrolyte and K i...