Ionic EquilibriumHard
Question
H3PO4 is a weak triprotic acid; approximate pH of 0.1 M Na2HPO4 (aq.) is calculated by :-
Options
A.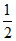 [pKa1+ pKa2]
[pKa1+ pKa2]
B.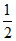 [pKa2 + pKa3]
[pKa2 + pKa3]
C.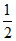 [pKa1 + pKa3]
[pKa1 + pKa3]
D.pKa1 + pKa2
More Ionic Equilibrium Questions
The pKa of a weak acid, HA, is 4.80. The pKb of a weak base, BOH, is 4.78. The pH of an aqueous solution of the correspo...For the titration of a dibasic weak acid H2A $\left( p^{K_{a(2)}} - p^{K_{a(1)}} \geq 2 \right)$ with a strong base, pH ...What is the difference in pH for 1/3 and 2/3 stages of neutralisation of 0.1 M CH3COOH with 0.1 M NaOH....Of the given anions, the strongest Bronsted base is :...A certain buffer solution contains equal concentration of X- and HX. The Kb for X- is 10-10. The pH of the buffer is :...