Ionic EquilibriumHard
Question
The pKa of a weak acid, HA, is 4.80. The pKb of a weak base, BOH, is 4.78. The pH of an aqueous solution of the corresponding salt, BA, will be
Options
A.9.58
B.4.79
C.7.01
D.9.22
Solution
It is a salt of weak acid and weak base
[H+] =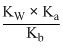
pH = 7.01
[H+] =
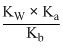
pH = 7.01
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
A sample of AgCl was treated with 5.00 ml of 2.0 M Na2CO3 solution to give Ag2CO3. The remaining solution contained 0.00...At 25oC, the solubility product of Mg(OH)2 is 1.0 × 10-11. At what pH, will Mg2+ ion start precipitating in the for...The self ionisation constant for pure formic acid, K = [HCOOH2+][HCOO-] has been estimated as 10-6 at room temperature. ...Ascorbic acid (vitamin C) is a diprotic acid, H2C6H6O6. What is the pH of a 0.10 M solution? The acid ionization constan...Biodegradable polymer which can be produced fromglycine and aminocaproic acid is :-...