Ionic EquilibriumHard
Question
What is the difference in pH for 1/3 and 2/3 stages of neutralisation of 0.1 M CH3COOH with 0.1 M NaOH.
Options
A.2 log 3
B.2 log (1/4)
C.2 log (2/3)
D.2 log 2
Solution
CH3COOH + OH- → CH3COO- + H2O
Difference in pH between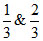 stages of neutralisation =
stages of neutralisation = 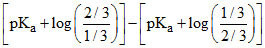 = 2 log 2
= 2 log 2
Difference in pH between
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The species present in solution when CO2 is dissolved in water are...A volume of 10 ml of a strong acid solution of pH = 2.0 are mixed with 990 ml of a buffer solution of pH = 4.0. The pH o...Which of the following relations is correct ?...Which of the following is the strongest base :-...Small amount of freshly precipitated magnesium hydroxides are stirred vigorously in a buffer solution containing 0.25 M ...