Ionic EquilibriumHard
Question
A certain buffer solution contains equal concentration of X- and HX. The Kb for X- is 10-10. The pH of the buffer is :
Options
A.4
B.7
C.10
D.14
Solution
According to Handerson′s equation
pH = pKα + log10
and Ka =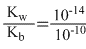 = 10-4, and [salt] = [Acid]
= 10-4, and [salt] = [Acid]
∴ pH = - log 10-10 + log10 = 4
= 4 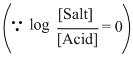
pH = pKα + log10

and Ka =
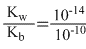 = 10-4, and [salt] = [Acid]
= 10-4, and [salt] = [Acid]∴ pH = - log 10-10 + log10
 = 4
= 4 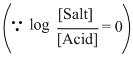
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The following graph represents the titration of pH vs volume Find out the best possible statement from the graph shown....The pH of a solution having the H+ ion concentration of 1 × 10-4 ions/litre is...Which of the following is true...What is the minimum mass of NaBr which should be added in 200 ml of 0.0004 M-AgNO3 solution just to start the precipitat...pH value of which of the following is not equal to one :...