Atomic StructureHard
Question
Actinium 231, 231Ac89, emit in succession two β-particles, four alphas one β and one alpha plus serveral γ rays. what is the resultant isotope :
Options
A.221Au79
B.211Au79
C.221Pb82
D.211Pb82
Solution
Five alpha and three beta particules results
231AC89 →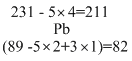
231AC89 →
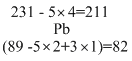
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
A radioactive material has half-life of 10 days. what fraction of the material would remain after 30 days?...Which on of the following is a possible nuclear reaction...During a negative beta decay...If an electron and a photon propagate in the form of waves having the same wavelength, it implies that they have the sam...Shown in the figure are two point charges +Q and - Q inside the cavity of a spherical shell. The charges arekept near th...