Ionic EquilibriumHard
Question
Which does not give iodoform reaction :-
Options
A.PhCOCH3
B.CH3OH
C.CH3CH2OH
D.CH3CHO
Solution
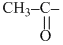 and
and 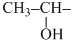 group containing compound gives iodoform test
group containing compound gives iodoform testCreate a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
The species present in solution when CO2 is dissolved in water are...The best indicator for detection of end point in titration of a weak acid and a strong base is :...or the equibrium CH3COOH(eq.) + H2O(l) ⇋ CH3COO-(aq.) + H3O+ (aq) ?Ka = 1.70 × 10-4 at 298 K. What is the eff...In which of the following cases, pH is greater than 7 ?...Solubility product constant (Ksp) of salts of types MX, MX2 and M3X at temperature ′T′ are 4.0 × 10-s, ...