Ionic EquilibriumHard
Question
Solubility product constant (Ksp) of salts of types MX, MX2 and M3X at temperature ′T′ are 4.0 × 10-s, 32 × 10-14 and 2.7 × 10-15, respectively. Solubilities (mole dm-3) of the salts at temperature ′T′ are in the order
Options
A.MX > MX2 > M3X
B.M3X > MX2 > MX
C.MX2 > M3X > MX
D.MX > M3X > MX2
Solution
Solubility of (MX) = 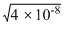 = 2 × 10-4
= 2 × 10-4
Solubility of (MX2) = 8 × 10-5
Solubility of (M3X) = 1 × 10-4
∴ MX > M3X > MX2
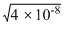 = 2 × 10-4
= 2 × 10-4Solubility of (MX2) = 8 × 10-5
Solubility of (M3X) = 1 × 10-4
∴ MX > M3X > MX2
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
What will be the resultant pH when 200 ml of an aqueous solution of HCl (pH = 2.0) is mixed with 300 ml of an aqueous so...H3A is a weak tribasic acid with Ka1 = 10−5, Ka2 = 10−9 and Ka3 = 10−13. The value of pX of 0.1 M – H3A solution, where ...At 25oC, the solubility product of Mg(OH)2 is 1.0 × 10-11. At what pH, will Mg2+ ion start precipitating in the for...Solid Ba (NO3)2 is gradually dissolved in a 1.0 × 10-4 M Na2CO3 solution. At what concentration ofBa2+ will a preci...The conjugate base of [AI(H2O)3(OH)3] is :...