Ionic EquilibriumHard
Question
In which of the following cases, pH is greater than 7 ?
Options
A.50 ml of 0.1 MHCl + 50 ml of 0.1 MNaCl
B.50 ml of 0.1 M H2SO4 + 50 ml of 0.2 M NaOH
C.50ml of 0.1 M CH3COOH + 50 ml of 0.1 M KOH
D.50 ml of 0.1 M HNO3 + 50 ml of 0.1 M NH3
Solution
(a) [H3O+] = 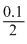 0.05;
0.05;
pH = - log 0.05 = 1.301
(b) Complete neturalization of strong acid and strong base, pH = 7.
(c) Hydrolysis of the salt CH3COOK, pH > 7 (salt of weak acid with strong base)
(d) Hydrolysis of the salt NH4NO3 pH < 7 (salt of strong acid with weak base)
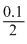 0.05;
0.05;pH = - log 0.05 = 1.301
(b) Complete neturalization of strong acid and strong base, pH = 7.
(c) Hydrolysis of the salt CH3COOK, pH > 7 (salt of weak acid with strong base)
(d) Hydrolysis of the salt NH4NO3 pH < 7 (salt of strong acid with weak base)
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Dissociation constants of acids HA and HB are 2.0 × 10−4 and 5 × 10−5, respectively. The [H+] in the resulting solution ...For the reaction [Ag(CN)2]– $\rightleftharpoons$ Ag+ + 2CN–, the equilibrium constant, at 25°C, is 4.0 × 10–19. Calcula...To 20 ml of 0.1 M – NaOH solution, 3 ml of 1 M acetic acid solution is added. Is the solution now neutral, acidic or alk...Which is most stable ?...To a 200 ml of 0.1 M weak acid HA solution 90 ml of 0.1 M solution of NaOH be added. Now, what volume of 0.1 M NaOH be a...