Chemical EquilibriumHard
Question
For NH4HS(s) ⇋ NH3(g) + H2S(g) ; observed pressure for reaction mixture at equilibrium is 1.12 atm at 106oC. The value of Kp for the reaction is :
Options
A.3.316 atm2
B.0.3136 atm2
C.31.36 atm2
D.6.98 atm2
Solution
NH4HS(s) ⇋ NH3(g) + H2S(g)
x + x = 1.12 atm
Kp =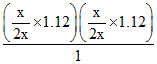
Kp = 0.3136 atm2
x + x = 1.12 atm
Kp =
Kp = 0.3136 atm2
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
For the reaction A + B$\rightleftharpoons$C + D, the initial concentration of A and B is equal, but the equilibrium conc...Solid ammonium carbamate dissociates as: NH2COONH4(s) $\rightleftharpoons$2NH3(g) + CO3(g). In a closed vessel, solid am...When $\text{NaNO}_3$ is heated in a closed vessel, oxygen is liberated and $\text{NaNO}_2$ is left behind. At equilibriu...XeF6 + H2O $\rightleftharpoons$ XeOF4 + 2HF; equilibrium constant = K1.XeO4 + XeF6 $\rightleftharpoons$ XeOF4 + XeO3F2; ...For the equilibrium SrCl2 · 6H2O(s) ⇋ SrCl2 · 2H2O(s) + 4H2O (g) the equilibrium constant KP = 16 × 10-12 atm...