Atomic StructureHard
Question
A diatomic molecule is made of two masses m1and m2 which are separated by a distance r. Ifwe calculate its rotational energy by applyingBohr′s rule of angular momentum quantization,its energy will be given by : (n is an integer)
Options
A.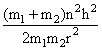
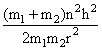
B.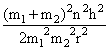
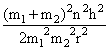
C.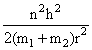
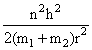
D.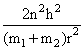
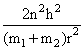
Solution
Energy 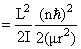
where μ = reduced mass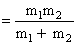
So Energy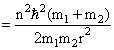
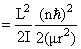
where μ = reduced mass
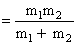
So Energy
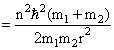
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
238U has 92 protons and 238 nucleons. It decays by emitting an Alpha particle and becomes...The wavelengths involved in the spectrum of deuterium (12D) are slightly different from that of hydrogen spectrum, becau...Binding energy per nucleon versus mass number curve for nuclei is shown in figure. W, X, Y and Z are four nuclei indicat...One mole of a monatomic ideal gas undergoes a cyclic process as shown in the figure (whre V is the volume and T is the t...Following are graphs of electric field variation with distance (r) for four different charge configurations.Choose the c...