Atomic StructureHard
Question
The half life of a radioactive substance against α - decay is 1.2 × 107 s. What is the decay rate for 4.0 × 1015 atoms of the substance
Options
A.4.6 × 1012 atoms/s
B.2.3 × 1011 atoms/s
C.4.6 × 1010 atoms/s
D.2.3 × 108 atoms/s
Solution
We have 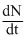 = λN
= λN
λ =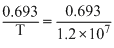
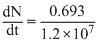 × 4 × 1015
× 4 × 1015
= 2.3 × 108 atom/s
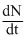 = λN
= λNλ =
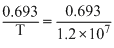
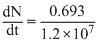 × 4 × 1015
× 4 × 1015 = 2.3 × 108 atom/s
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Electrons used in an electron microscope are accelerated by a voltage of 25 kV. If the voltage is increased to 100 kV th...When photo energy 4.25 eV strike the surface of a metal A, the ejected photoelectrons have maximum kinetic energy TA exp...A diatomic molecule is made of two masses m1and m2 which are separated by a distance r. Ifwe calculate its rotational en...The wavelength λe of an electron and λp of a photon of same energy E are related by:...Energy equivalent to 10gm of U235 is: -...