Atomic StructureHard
Question
A photon of 1.7 × 10-13 Joules is absorbed by a material under special circumstances. The correct statement is :-
Options
A.Electrons of the atom of absorbed material will go the higher energy states
B.Electron and positron pair will be created
C.Only positron will be produced
D.Photoelectric effect will occur and electron will be produced
Solution
Energy = 1.7 × 10-13 J
=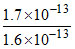 MeV = 1.0625 MeV
MeV = 1.0625 MeV
⇒ Pair production will take place
=
⇒ Pair production will take place
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The de-Broglie wavelength associated with the electron in the n = 4 level is :...A hydrogen atom and a $\mathrm{Li}^{2+}$ ion are both in the second excited state. If $l_H$ and $l_{Li}$ are their respe...A full wave rectifier circuit along with the output is shown in figure. The contribution (s) from the diode 1 is (are)...A radioactive material decays by simultaneous emission of two particles with respective half lives 1620 and 810 years. T...Two hydrogen atoms are in excited state with electrons residing in n = 2. First one is moving towards left and emits a p...