ThermodynamicsHard
Question
p-V plots for two gases during adiabatic processes are shown in the figure. Plots 1 and 2 should correspond respectively to
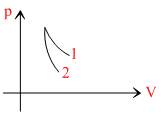

Options
A.He and O2
B.O2 and He
C.He and Ar
D.O2 and N2
Solution
In adiabatic process
slope of p-V graph,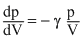
slope ∝ γ [with negative sign]
From the given graph,
(slope)2 > (slope)1
∴ γ2 > γ1
Therefore, 1 should corresponding to O2 (γ = 1.14) and 2 should correspond to He (γ = 1.67).
slope of p-V graph,
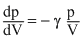
slope ∝ γ [with negative sign]
From the given graph,
(slope)2 > (slope)1
∴ γ2 > γ1
Therefore, 1 should corresponding to O2 (γ = 1.14) and 2 should correspond to He (γ = 1.67).
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The average translational energy and the rms speed of molecules in a sample of oxygen gas at 300 K are 6.21 × 10-21...Two moles of ideal helium gas are in a rubber balloon at 30oC. The balloon is fully expandable and can be assumed to req...Two moles of helium gas are taken over the cycle ABCDA, as shown in the P - T diagram. The net work done on the gas in t...One mole of an ideal atomic gas is heated at a constant pressure of one atmosphere from 0oC to 100oC. Then the change in...Two bodies A and B have thermal emissivities of 0.01 and 0.81 respectively. The outer surface area of the two bodies are...