ThermodynamicsHard
Question
Two moles of helium gas are taken over the cycle ABCDA, as shown in the P - T diagram.
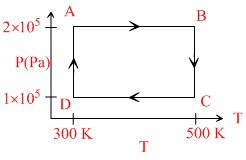
The net work done on the gas in the cycle ABCDA is

The net work done on the gas in the cycle ABCDA is
Options
A.Zero
B.276 R
C.1076 R
D.1904 R
Solution
Net work done in a cycle = WAB + WBC + WCB + WBA
= 400 R + 2 × 2.303 × 500 R ln 2 - 400R - 414 R
= 1000R x ln 2 - 600R x ln 2 = 400R x ln 2 = 276R
= 400 R + 2 × 2.303 × 500 R ln 2 - 400R - 414 R
= 1000R x ln 2 - 600R x ln 2 = 400R x ln 2 = 276R
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The absolute zero is the temperature at which:...In the given figure graph is drawn between and P at different temperature, where n is the numberof moles and R is univer...Air is filled in a tube of the wheel of a car at 27o C and 2 atm pressure if the tube is suddenly bursts, the final temp...An ideal gas is subjected to an isothermal expansion such that its volume change fromVi to Vf and pressure from Pi to Pf...100g of water is heated from 30oC to 50oC. Ignoring the slight expansion of the water, the change in its internal energy...