ThermodynamicsHard
Question
The average translational energy and the rms speed of molecules in a sample of oxygen gas at 300 K are 6.21 × 10-21 J and 484 m/s respectively. The corresponding values at 600 K are nearly (assuming ideal gas behaviour)
Options
A.12.42 × 10-21 J, 968 m/s
B.8.78 × 10-21 J, 684 m/s
C.6.21 × 10-21 J, 968 m/s
D.12.42 × 10-21 J, 684 m/s
Solution
The average translation KE = 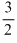 kT which is directly proportional to T, while rms speed of molecules is given by
kT which is directly proportional to T, while rms speed of molecules is given by
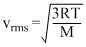 i.e., vrms 0 ∝ √T
i.e., vrms 0 ∝ √T
When temperature of gas is increased from 300 K to 600 K (i.e., 2 times), the average translation KE will increase to 2 times and rms speed to √2 or 1.414 times.
∴ Average translational KE = 2 × 6.21 × 10-21 J
= 12.42 × 10-21 J
and vrms = (1.414) (484) m/s
 684 m/s
684 m/s
 kT which is directly proportional to T, while rms speed of molecules is given by
kT which is directly proportional to T, while rms speed of molecules is given by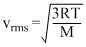 i.e., vrms 0 ∝ √T
i.e., vrms 0 ∝ √TWhen temperature of gas is increased from 300 K to 600 K (i.e., 2 times), the average translation KE will increase to 2 times and rms speed to √2 or 1.414 times.
∴ Average translational KE = 2 × 6.21 × 10-21 J
= 12.42 × 10-21 J
and vrms = (1.414) (484) m/s
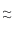 684 m/s
684 m/sCreate a free account to view solution
View Solution FreeMore Thermodynamics Questions
In the given figure graph is drawn between and P at different temperature, where n is the numberof moles and R is univer...The process of converting solid into directly gaseous state on being heated is called :-...A vessel contains a mixture of one mole of oxygen and two mole of nitrogen at 300 K. The ratio of the average rotational...For an ideal gas, each of the following processes definitely leads to an change in the pressure of the gas, except one. ...A gas at 50 N/m2 pressure is compressed from 10m3 to 4m3 under constant pressure. subsequently it is given 100 J of heat...